Background
Project ID: D2010-06
Invention novelty:
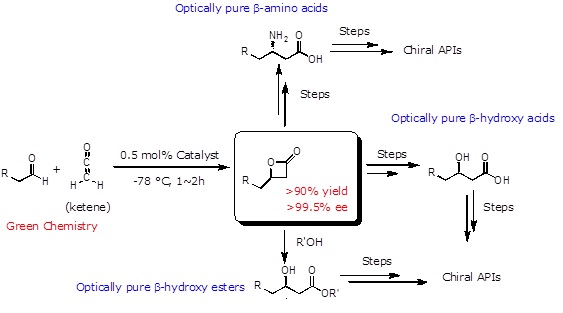
The disclosed invention embodies a novel cobalt based bifunctional catalyst and the process to synthesize enantiomerically pure ( > 99 % ee) β-lactones.
Value proposition:
Enantiomerically pure β-lactones have found attraction as intermediates in the synthesis of a large number of chiral drugs. However, the costly and time consuming methods to synthesize β-lactones and the impure forms of β-lactones so produced pose a big challenge to the pharmaceutical industry. The present invention sets off these limitations and provides novel cobalt (III) based Lewis acid-Lewis base bifunctional catalyst and methods for producing optically pure β-lactones. The advantages include:
- Production of enantiomerically pure (>99 %) C4-alkyl or aryl β-lactones.
- Unprecedented reaction time and efficiency.
- High reaction rate acceleration and perfect enantioselectivity.
Invention Description:
Enantiomerically pure β-lactones are valuable small molecules that can be converted into a variety of chiral synthons for asymmetric synthesis. Because the [2+2] cycloaddition reaction between ketene and aldehydes affords β-lactones in one step, developing new catalytic methods for synthesizing β-lactones from ketene and aldehydes attracts much attention.
A researcher at the University of Toledo has developed a novel tethered Lewis acid-Lewis base bifunctional catalyst, by covalently linking a metallosalen Lewis acid to a cinchona alkaloid Lewis base. The resulting Lewis acid-Lewis base bifunctional catalyst displays remarkable reaction rate acceleration through the cooperative dual activations of the two reacting partners organized intramolecularly by the catalyst. This catalyst promotes the rapid formation of optically pure β-lactones from aldehydes and unsubstituted ketene, with reaction times ranging from mere minutes to an hour or less.
Schematic:
Applications:
Synthesis of highly valuable chiral molecules in biotechnological, chemical, and pharmaceutical industries.
IP Status: 9,334,266